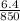Physics, 22.06.2019 21:00 tobyfoerst
If the specific heat of a metal is 0.850 j/g °c, what is its atomic weight?

Answers: 2


Other questions on the subject: Physics



Physics, 23.06.2019 12:00, bks53
Unlike the idealized voltmeter, a real voltmeter has a resistance that is not infinitely large. a voltmeter with resistance rv is connected across the terminals of a battery of emf e and internal resistance r. find the potential difference vmeter measured by the voltmeter. what is vmeter?
Answers: 2

Physics, 23.06.2019 16:20, SmokeyRN
Amagnetically soft material is placed in a strong magnetic field. what is the most likely outcome? it will become a permanent magnet because the domains will remain aligned. it will become a temporary magnet because the domains will remain aligned. it will become a permanent magnet because the domains will easily realign. it will become a temporary magnet because the domains will easily realign.
Answers: 2
You know the right answer?
If the specific heat of a metal is 0.850 j/g °c, what is its atomic weight?...
Questions in other subjects:

Biology, 19.11.2019 19:31

Mathematics, 19.11.2019 19:31








English, 19.11.2019 19:31

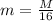
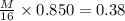
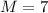
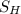
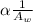
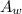 is the atomic weight of the object.
is the atomic weight of the object.