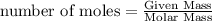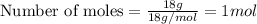Physics, 29.06.2019 22:00 jaidenkenna2001
Specific heat of water = 4.186 j g°c specific heat of ice = 2.00 j g°c molar heat of fusion = 6030 j mol molar heat of vaporization = 40790 j mol you take an ice cube (mass = 18g) from the freezer (t = -10°c) and place it on the table. later that day, you notice a puddle of water on the table that has reached ambient room temperature (20°c). how much heat must have been absorbed to make this happen?

Answers: 1


Other questions on the subject: Physics

Physics, 22.06.2019 14:30, Paigex3
In order to do work, the force vector must be question 1 options: in a different direction than the acceleration vector. in a different direction than the displacement vector. in the same direction as the displacement vector and the motion. in the same direction as the acceleration vector.
Answers: 1


You know the right answer?
Specific heat of water = 4.186 j g°c specific heat of ice = 2.00 j g°c molar heat of fusion = 6030...
Questions in other subjects: