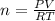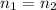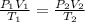Physics, 14.07.2019 16:30 keaton3410
According to avogadro's law, the volume of a gas is inversely related to the number of moles at constant temperature and pressure the volume of a gas is inversely related to the number of moles at standard temperature and pressure the volume of a gas depends only on the temperature and pressure the volume of a gas depends only on the number of moles in the sample the volume of a gas is directly related to the number of moles at constant temperature and pressure

Answers: 1


Other questions on the subject: Physics

Physics, 21.06.2019 14:50, Theresab2021
Ingrid wrote the hypothesis below. if the temperature of a liquid increases, the density of the liquid decreases because the particles move farther apart. what are the variables in her hypothesis?
Answers: 3

Physics, 21.06.2019 22:50, sarahfsakrani
Aplane takes off from an airport and flies to town a, located d1 = 215 km from the airport in the direction 20.0° north of east. the plane then flies to town b, located d2 = 230 km at 30.0° west of north from town a. use graphical methods to determine the distance and direction from town b to the airport. (enter the distance in km and the direction in degrees south of west.)
Answers: 2

Physics, 22.06.2019 09:20, Geo777
Question 11 of 13 (1 point) jump to question: a stairway, ladder, or ramp must be present in excavations in which of the following situations? a. all trenches must have access/egress b. the trench is more than 15 feet wide but only 1 ½ feet deep c. the trench is more than 4 feet deep and the devices must be within 25 feet of all workers
Answers: 1
You know the right answer?
According to avogadro's law, the volume of a gas is inversely related to the number of moles at con...
Questions in other subjects:

Chemistry, 05.02.2021 04:40


Mathematics, 05.02.2021 04:40

Mathematics, 05.02.2021 04:40





English, 05.02.2021 04:40