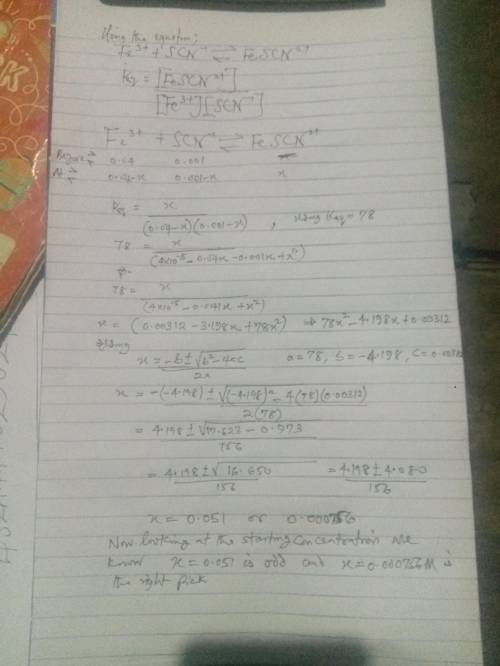Mathematics, 11.02.2020 21:27 Student2499
Using your calculated average value for Keq , calculate the SCN- concentration in a solution with an initial Fe3+ concentration of 4.00 x 10-2 M and the initial concentration of SCN- was 1.00 x 10-3 M. Is all the SCN- ion in the form of FeSCN2+? Hint: Set your product concentration to "x" and use the quadratic equation to solve. You will need to rearrange your Keq equation into the form of ax2 + bx + c = 0

Answers: 2


Other questions on the subject: Mathematics



You know the right answer?
Using your calculated average value for Keq , calculate the SCN- concentration in a solution with an...
Questions in other subjects:

English, 29.12.2019 09:31


Mathematics, 29.12.2019 09:31


Mathematics, 29.12.2019 09:31

Social Studies, 29.12.2019 09:31

Mathematics, 29.12.2019 09:31

Computers and Technology, 29.12.2019 09:31

English, 29.12.2019 09:31