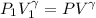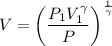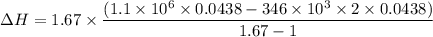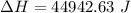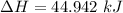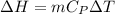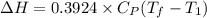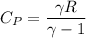Engineering, 25.09.2020 04:01 khliyahmccaskil
Dr. Thermo, only has one bottle of neon. However, he needs to run two experiments, each requiring its own bottle. Therefore, he plans to connect the two bottles together and open the valves on each so that each bottle is partially filled. He wants to know how the enthalpy of the gas will change when he performs this operation. Each bottle has an internal volume of 43.8 L, is completely rigid, and fully insulated. At the start, the full bottle has a pressure of 1.1 MPa, the second bottle is completely evacuated, and both are at room temperature (298 K). After the valves are opened, the two bottles come to equilibrium at 346 kPa. You can assume that neon behaves ideally during this process.
a. Dr. Thermo wants you to derive an equation for H(P. V) and then use that equation to determine the change in enthalpy by integration, showing him all your work.
b. Being a thermo wiz, you know there is another (and easier) way to perform this calculation. Verify your answer to part a using this easier way.

Answers: 3


Other questions on the subject: Engineering

Engineering, 03.07.2019 14:10, volleyballfun24
If the thermal strain developed in polyimide film during deposition is given as 0.0044. assume room temperature is kept at 17.3 c, and thermal coefficient of expansion for the film and the substrate are 54 x 10^-6c^-1 and 3.3 x 10^-6c^-1respectively. calculate the deposition temperature.
Answers: 3

Engineering, 04.07.2019 18:10, jadeochoa4466
The temperature of air decreases as it is compressed by an adiabatic compressor. a)- true b)- false
Answers: 2

Engineering, 04.07.2019 18:10, hadellolo8839
Acompressor receives the shaft work to decrease the pressure of the fluid. a)- true b)- false
Answers: 3
You know the right answer?
Dr. Thermo, only has one bottle of neon. However, he needs to run two experiments, each requiring it...
Questions in other subjects:




Arts, 23.10.2020 16:10


Chemistry, 23.10.2020 16:10

Arts, 23.10.2020 16:10

Spanish, 23.10.2020 16:10

Mathematics, 23.10.2020 16:10

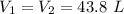
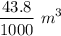
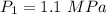 and
and 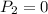

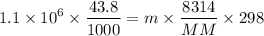
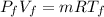
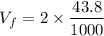
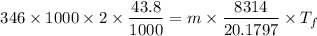
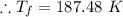
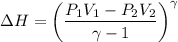 as
as 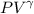 = constant
= constant