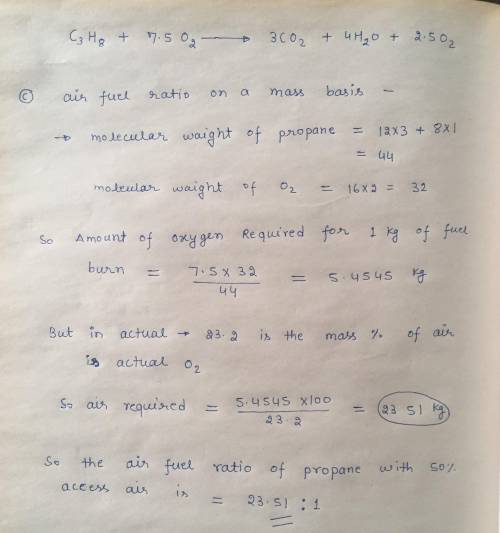Engineering, 06.05.2020 02:09 dorindaramirez0531
Propane gas C3H8 enters a combustion chamber operating at steady state condition at 1 bar, 25ºC and is burned with 150% theoretical air that enters the combustion chamber at the same state. The products leave the combustion chamber at 1 bar and 1200 K.
a) Write down the balanced chemical equation for the complete combustion.
b) Write down the balanced actual combustion reaction and calculate c) the air fuel ratio on a mass basis, d) the dew point temperature of the products in ºC, e) the heat transfer in terms of kJ/kmol of fuel.

Answers: 2


Other questions on the subject: Engineering

Engineering, 03.07.2019 15:10, breannaasmith1122
Two flowing streams of argon gas are adiabatically mixed to form a single flow/stream. one stream is 1.5 kg/s at 400 kpa and 200 c while the second stream is 2kg/s at 500 kpa and 100 ? . it is stated that the exit state of the mixed single flow of argon gas is 150 c and 300 kpa. assuming there is no work output or input during the mixing process, does this process violate either the first or the second law or both? explain and state all your assumptions.
Answers: 1


Engineering, 04.07.2019 18:20, safiyabrowne7594
Ahe-xe mixture containing a 0.75 mole fraction of helium is used for cooling electronics in an avionics application. at a temperature of 300 k and atmospheric pressure, calculate the mass fraction of helium and the mass density, molar concentration and molecular weight of the mixture. if the cooling capacity is 10 l, what is the mass of the coolant?
Answers: 3
You know the right answer?
Propane gas C3H8 enters a combustion chamber operating at steady state condition at 1 bar, 25ºC and...
Questions in other subjects:

Mathematics, 08.12.2020 02:40

English, 08.12.2020 02:50

Mathematics, 08.12.2020 02:50

Mathematics, 08.12.2020 02:50


Health, 08.12.2020 02:50



Mathematics, 08.12.2020 02:50

Mathematics, 08.12.2020 02:50