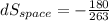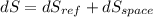Engineering, 14.04.2020 21:41 kenzie3497
Refrigerant-134a enters the coils of the evaporator of a refrigeration system as a saturated liquid–vapor mixture at a pressure of 140 kPa. The refrigerant absorbs 180 kJ of heat from the cooled space, which is maintained at -10oC, and leaves as saturated vapor at the same pressure. Determine (a) the entropy change of the refrigerant, (b) the entropy change of the cooled space, and (c) the total entropy change for this process.

Answers: 3


Other questions on the subject: Engineering

Engineering, 04.07.2019 03:10, lauriepdx17
What precautions should you take to prevent injuries when dealing with heavy loads?
Answers: 1

Engineering, 04.07.2019 18:10, heidiburgos1own6c0
Fluids at rest possess no flow energy. a)- true b)- false
Answers: 3

Engineering, 04.07.2019 18:10, salazjlove
Which of the following refers to refers to how well the control system responds to sudden changes in the system. a)-transient regulation b)- distributed regulation c)-constant regulation d)-steady-state regulation
Answers: 1

Engineering, 04.07.2019 18:20, cristykianpour
Describe one experiment in which the glass transition temperature and melting temperature of a totally amorphous thermoplastic material can be determined. show the relevant experimental results in a diagram which should be properly annotated with the two temperatures clearly marked. what is likely to happen to the curve in the diagram if the amorphous polymer is replaced by a thermosetting type?
Answers: 2
You know the right answer?
Refrigerant-134a enters the coils of the evaporator of a refrigeration system as a saturated liquid–...
Questions in other subjects:

Chemistry, 21.10.2020 14:01


Social Studies, 21.10.2020 14:01

Chemistry, 21.10.2020 14:01

Biology, 21.10.2020 14:01

English, 21.10.2020 14:01

Chemistry, 21.10.2020 14:01



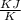
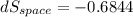
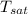 = - 18.77 °c = - 18.77 + 273 = 254.23 K
= - 18.77 °c = - 18.77 + 273 = 254.23 K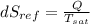
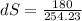
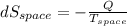
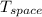 = - 10 °c = 263 K
= - 10 °c = 263 K