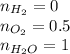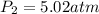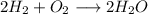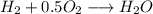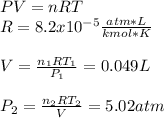Engineering, 02.09.2019 16:30 ineedhelp368
Amixture of 1 kmol of hydrogen (h2) and n kmol of oxygen (o2), initially at 25°c and 1 atm, burns completely in a closed, rigid, insulated container. the container finally holds a mixture of water vapor and o2 at 2000 k. the ideal gas model applies to each mixture and there is no change in kinetic or potential energy between the initial and final states. determine: (a) the value of n. (b) the final pressure, in atm.

Answers: 3


Other questions on the subject: Engineering

Engineering, 03.07.2019 14:10, kayabwaller4589
When at a point two solid phase changes to one solid phase on cooling then it is known as a) eutectoid point b) eutectic point c) peritectic point d) peritectoid point
Answers: 3

Engineering, 04.07.2019 03:10, lauriepdx17
What precautions should you take to prevent injuries when dealing with heavy loads?
Answers: 1

Engineering, 04.07.2019 18:10, alyssabailey7545
Give heat transfer applications for the following, (i) gas turbines (propulsion) ) gas turbines (power generation). (iii) steam turbines. (iv) combined heat and power (chp). (v) automotive engines
Answers: 1

Engineering, 04.07.2019 18:10, katelynn73
Atmospheric air has a temperature (dry bulb) of 80° f and a wet bulb temperature of 60° f when the barometric pressure is 14.696 psia. determine the specific humidity, grains/lb dry air. a. 11.4 c. 55.8 d. 22.5 b. 44.1
Answers: 1
You know the right answer?
Amixture of 1 kmol of hydrogen (h2) and n kmol of oxygen (o2), initially at 25°c and 1 atm, burns co...
Questions in other subjects:

History, 16.10.2020 09:01





Social Studies, 16.10.2020 09:01



Chemistry, 16.10.2020 09:01