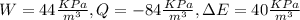Engineering, 24.08.2019 02:10 sarakiker
Determine which of the following sets of data satisfy the first law of thermodynamics? (recall that e = total energy = u + ke + pe). a. w = 50 kj q = 170 kj deltae = 120 kj b. w = 100 btu q = -110 btu deltae = -210 btu c. w = 250 kj q = -110 kj deltae = -100 wh d. w = 44 kpa-m3 q = -84 kpa-m3 delate = 40 kn-m

Answers: 3


Other questions on the subject: Engineering

Engineering, 04.07.2019 18:10, viicborella
Steel is coated with a thin layer of ceramic to protect against corrosion. what do you expect to happen to the coating when the temperature of the steel is increased significantly? explain.
Answers: 1


Engineering, 04.07.2019 18:10, agpraga23ovv65c
Carbon dioxide gas expands isotherm a turbine from 1 mpa, 500 k at 200 kpa. assuming the ideal gas model and neglecting the kinetic and potential energies, determine the change in entropy, heat transfer and work for each kilogram of co2.
Answers: 2

Engineering, 04.07.2019 18:10, wyattlb97
Water at the rate of 1 kg/s is forced through a tube with a 2.5 cm inner diameter. the inlet water temperature is 15°c, and the outlet water temperature is 50°c. the tube wall temperature is 14°c higher than the local water temperature all along the length of the tube. what is the length of the tube?
Answers: 3
You know the right answer?
Determine which of the following sets of data satisfy the first law of thermodynamics? (recall that...
Questions in other subjects:

Chemistry, 28.02.2021 19:00


English, 28.02.2021 19:00