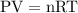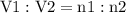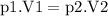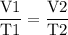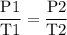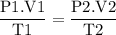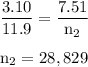Chemistry, 22.07.2019 19:00 alejandra340
Aflexible container at an initial volume of 3.10 l contains 7.51 mol of gas. more gas is then added to the container until it reaches a final volume of 11.9 l. assuming the pressure and temperature of the gas remain constant, calculate the number of moles of gas added to the container.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, tbiles99
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 14:00, leahstubbs
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 23.06.2019 07:00, asims13
The following transition occurs at a molecular level for a substance. what transition corresponds to this change in microscopic structure? the carbon dioxide molecules on the left are in a regular, tightly packed pattern. after heating, it becomes much lower density. a. melting b. boiling c. sublimation d. freezing
Answers: 1

Chemistry, 23.06.2019 11:00, alejachico2
Suppose you increase your walking speed from 7 m/s to 15 m/s in a period of 1 s. what is your acceleration?
Answers: 1
You know the right answer?
Aflexible container at an initial volume of 3.10 l contains 7.51 mol of gas. more gas is then added...
Questions in other subjects:






Arts, 10.12.2020 19:50

Mathematics, 10.12.2020 19:50

Mathematics, 10.12.2020 19:50

Mathematics, 10.12.2020 19:50