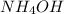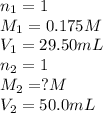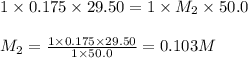Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, kkruvc
Will mark brainliest 26. which of these statements are true? (3 points) a. gases are compressible b. gases fill their containers completely c. the pressure of a gas is independent of the temperature d. gases have mass e. gases exert pressure f. the pressure of a gas is dependent on the volume g. gas pressure results from the collisions between gas particles h. gases have a definite volume and shape
Answers: 1


Chemistry, 22.06.2019 20:00, Isaiahtate053
The volume of a single vanadium atom is 9.29×10-24 cm3. what is the volume of a vanadium atom in microliters?
Answers: 3

Chemistry, 22.06.2019 21:00, agarcia24101993
What is the chemical formula for the compound formed between sodium and flour one
Answers: 1
You know the right answer?
If 29.50 ml of 0.175m nitric acid neutralizes 50.0 ml of ammonium hydroxide, what is the molarity of...
Questions in other subjects:

Chemistry, 02.09.2019 17:50


Social Studies, 02.09.2019 17:50

Mathematics, 02.09.2019 17:50

English, 02.09.2019 17:50

Biology, 02.09.2019 17:50



Social Studies, 02.09.2019 17:50

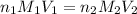
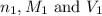 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 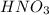
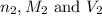 are the n-factor, molarity and volume of base which is
are the n-factor, molarity and volume of base which is