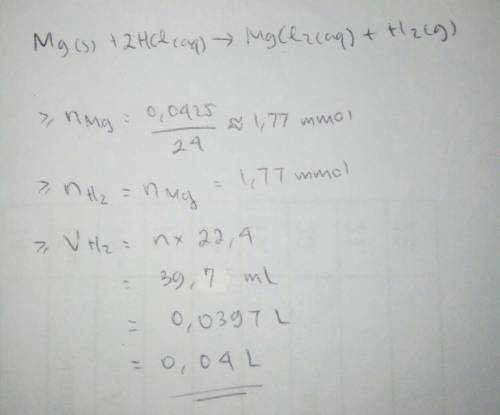Chemistry, 23.07.2019 04:00 jdkrisdaimcc11
If 0.04250g of magnesium is reacted with an excess of hydrochloric acid, calculate the theoretical volume at stp of hydrogen gas produced.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:00, ahnorthcutt4965
Acm ruler with main graduations from 1 to 10 from left to right there are 10 secondary graduations between each of the main graduations there is a line that begins. at the left end of the ruler 10 secondary graduations to the left of the “1 main graduation the right end of the line ends on the eighth secondary graduation to the right of 3 how long is the line
Answers: 1

Chemistry, 22.06.2019 22:30, xlebrny7831
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2

Chemistry, 23.06.2019 00:30, motorxr714
Titration reveals that 11.6 ml of 3.0m sulfuric acid are required to neutralize the sodium hydroxide in 25.00ml of naoh solution. what is the molarity of the naoh solution?
Answers: 1
You know the right answer?
If 0.04250g of magnesium is reacted with an excess of hydrochloric acid, calculate the theoretical v...
Questions in other subjects:

Biology, 26.07.2019 00:40


History, 26.07.2019 00:40


Mathematics, 26.07.2019 00:40

Physics, 26.07.2019 00:40

Mathematics, 26.07.2019 00:40


Biology, 26.07.2019 00:40