Chemistry, 25.07.2019 08:30 itcelmairani
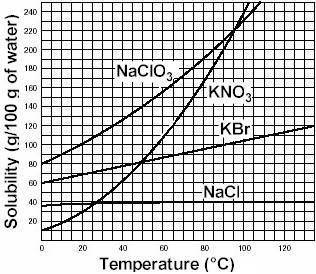
Asolution is a homogeneous mixture of one or more solutes dissolved in a solvent. a specific volume of solvent is only able to dissolve a limited amount of solute. as long as the solvent is able to dissolve more solute, the solution remains unsaturated. when the solvent can no longer dissolve additional solute, the solution is saturated. at this point, any additional solute will fall to the bottom of the container. the amount of solute that can be dissolved in a given amount of solvent at a specific temperature and pressure is defined as the solubility of the solute. consider the solubility curves of several salts in water. based on the information here, if 220 grams of the salt kbr are added to 100 ml of water at 100oc, we would label that solution as a) insoluble b) saturated c) supersaturated d) unsaturated

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:30, CoolRahim9090
Which of the following represents the ester functional group? a. -coo- b. -cho c. -cooh d. c=o
Answers: 1


Chemistry, 22.06.2019 19:10, krisandlance
Astudent completes a titration by adding 12.0 milliliters of naoh(aq) of unknown concentration to 16.0 milliliters of 0.15 m hcl(aq). what is the molar concentration of the naoh(aq)? 1)5.0 m 2)0.20 m 3)0.11 m 4)1.1 m
Answers: 1
You know the right answer?
Asolution is a homogeneous mixture of one or more solutes dissolved in a solvent. a specific volume...
Questions in other subjects:


Physics, 11.07.2019 17:30

History, 11.07.2019 17:30

English, 11.07.2019 17:30

Mathematics, 11.07.2019 17:30

Business, 11.07.2019 17:30



Mathematics, 11.07.2019 17:30