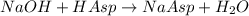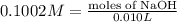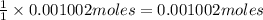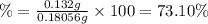Chemistry, 26.07.2019 03:30 codyshs160
Astudent found that the titration had taken 10.00 ml of 0.1002 m naoh to titration 0.132 g of aspirin, a monoprotic acid. calculate the percent purity of aspirin (c9h8o4, molar mass = 180.2 g/mol) sample.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:30, hellokitty1647
For the following dehydrohalogenation (e2) reaction, draw the zaitsev product(s) resulting from elimination involving c3–c4 (i. e., the carbon atoms depicted with stereobonds). show the product stereochemistry clearly. if there is more than one organic product, both products may be drawn in the same box. ignore elimination involving c3 or c4 and any carbon atom other than c4 or c3.
Answers: 3


You know the right answer?
Astudent found that the titration had taken 10.00 ml of 0.1002 m naoh to titration 0.132 g of aspiri...
Questions in other subjects:


Biology, 10.03.2020 18:12

Mathematics, 10.03.2020 18:12





English, 10.03.2020 18:12