
Which characteristics of metal atoms explain why valence electrons in a metal are delocalized? metal atoms are large and have high electronegativities. metal atoms are small and have low electronegativities. metal atoms are large and have low electronegativities. metal atoms are small and have high electronegativities.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:00, melissa9882
A50.0g sample of liquid water at 0.0 c ends up as ice at -20.0 c. how much energy is involved in this change?
Answers: 1

Chemistry, 22.06.2019 17:00, brownvester44
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2

Chemistry, 23.06.2019 02:00, Turtlelover05
Pinene is an unsaturated hydrocarbon found in pine resin. if pinene has m+ = 136 and contains 1 double bond(s) and 2 ring(s); what is its molecular formula? enter the formula in the form ch first, then all other atoms in alphabetical order; do not use subscripts. the formula is case-sensitive
Answers: 3
You know the right answer?
Which characteristics of metal atoms explain why valence electrons in a metal are delocalized? met...
Questions in other subjects:


Mathematics, 06.10.2019 10:00

Mathematics, 06.10.2019 10:00

English, 06.10.2019 10:00





History, 06.10.2019 10:00

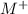 ions and group II elements are alkaline earth metals, they can give 2 electrons to form
ions and group II elements are alkaline earth metals, they can give 2 electrons to form 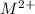 ions.
ions.

