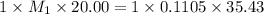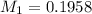You are given a solution of hcooh (formic acid) with an approximate concentration of 0.20 m and you will titrate this with a 0.1105 m naoh. you add 20.00 ml of hcooh to the beaker before titrating, and it requires 35.43 ml of naoh to reach the end point. what is the concentration of the hcooh solution?

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 10:10, dhailyortegacampa131
Stage in which a typical star has completely stopped fusion
Answers: 1

Chemistry, 22.06.2019 17:00, brandiwingard
What is the mass of phosphorous in a 51-kg person
Answers: 1
You know the right answer?
You are given a solution of hcooh (formic acid) with an approximate concentration of 0.20 m and you...
Questions in other subjects:





Computers and Technology, 25.11.2021 06:40

Mathematics, 25.11.2021 06:40


Mathematics, 25.11.2021 06:40

English, 25.11.2021 06:40


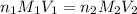
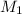 = molarity of
= molarity of 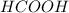 solution = ?
solution = ?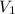 = volume of
= volume of 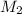 = molarity of
= molarity of 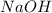 solution = 0.1105 M
solution = 0.1105 M
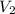 = volume of
= volume of 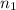 = valency of
= valency of 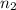 = valency of
= valency of