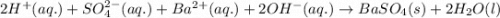Chemistry, 28.07.2019 18:00 claytonashley30
Identify the spectator ion in this reaction. highlight your answer. ba(oh)2(aq) + h2so4(aq) → baso4(s) + h2o(l) ba2+ so42- oh- h+ there is no spectator ion.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:00, misspicafunpoke
Indicate whether the specified alkyl halides will form primarily substitution products, only elimination products, both substitution and elimination products, or no products when they react with sodium methoxide. 1-bromobutane 1-bromo-2-methylpropane 2-bromobutane 2-bromo-2-methylpropane
Answers: 2

Chemistry, 22.06.2019 12:10, purplefish53
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3


Chemistry, 22.06.2019 19:30, gracieisweird12
Use the periodic table to find the molar mass of each element. molar mass h = g/mol molar mass s = g/mol molar mass o = g/mol
Answers: 3
You know the right answer?
Identify the spectator ion in this reaction. highlight your answer. ba(oh)2(aq) + h2so4(aq) → baso4(...
Questions in other subjects:


Social Studies, 01.09.2019 17:30


Mathematics, 01.09.2019 17:30

English, 01.09.2019 17:30

Mathematics, 01.09.2019 17:30

Chemistry, 01.09.2019 17:30



Biology, 01.09.2019 17:30