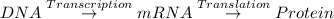Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 13:00, willcohen42
What postulate of the kinetic molecular theory best explains why gases have high fluidity? because collisions between gas particles are elastic, there is no loss of energy as particles flow past each other. because gases consist of large numbers of tiny particles, they spread out and do not come in contact with each other. because the attractive forces between gas particles are negligible, gas particles can glide easily past one another. because the average kinetic energy of gas particles increases as temperature increases, gas particles behave more like a liquid. question 6 compare the compressibility of gases and liquids. support your answer by describing the arrangement of particles in gases and liquids.
Answers: 1


Chemistry, 22.06.2019 16:10, 00015746
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 23.06.2019 01:00, stefaniethibodeaux
Substance 33°f 100°f peanut oil solid liquid margarine solid liquid chocolate chips solid liquid which conclusion fits the data in the table? a. heat chemically changes chocolate and margarine. b. all solids become liquid at 100°f. c. removing heat from a substance it to melt. d. matter may change shape when it is heated.
Answers: 1
You know the right answer?
What does mrna copy and carry the code to produce? a. dna b. proteins c. gametes d. organelles...
Questions in other subjects:

Medicine, 19.07.2019 02:20

Engineering, 19.07.2019 02:20

Medicine, 19.07.2019 02:20

Computers and Technology, 19.07.2019 02:20

Computers and Technology, 19.07.2019 02:20


Medicine, 19.07.2019 02:20

Computers and Technology, 19.07.2019 02:20

Medicine, 19.07.2019 02:20

Computers and Technology, 19.07.2019 02:20