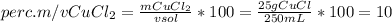Chemistry, 29.07.2019 13:00 abigaleschwartz
Dissolving 25.00 g cucl2 in enough water to make 250 ml of solution gives a solution concentration equal to

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, MickeyxX7096
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 16:00, jrocklove7825
About 3% of the water on earth is freshest. only about 40% of that freshwater is available for human use. why is so much freshwater unavailable for human use?
Answers: 2

Chemistry, 22.06.2019 16:00, julesperez22
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3
You know the right answer?
Dissolving 25.00 g cucl2 in enough water to make 250 ml of solution gives a solution concentration e...
Questions in other subjects:



Physics, 05.12.2019 18:31


Arts, 05.12.2019 18:31


Social Studies, 05.12.2019 18:31


Mathematics, 05.12.2019 18:31