
Chemistry, 05.10.2019 03:50 kolbehoneyman
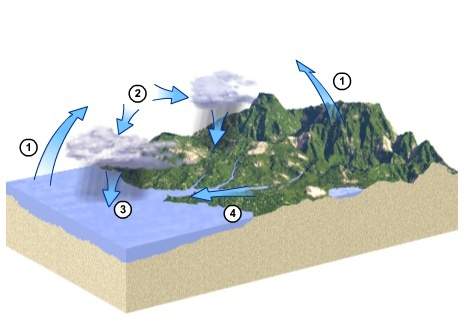
Which is represented by the arrow labeled 4 in the diagram? a. water is absorbing heat energy and changing from a liquid to a gas. b. water is releasing heat energy and changing from a gas to a liquid. c. water is running and collecting along the surface. d. water is being absorbed into the ground.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:00, coco8560
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 21:40, fatherbamboo
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3

You know the right answer?
Which is represented by the arrow labeled 4 in the diagram? a. water is absorbing heat energy and c...
Questions in other subjects:

Mathematics, 16.02.2021 17:40

Mathematics, 16.02.2021 17:40



Computers and Technology, 16.02.2021 17:40


Mathematics, 16.02.2021 17:40

Mathematics, 16.02.2021 17:40

Chemistry, 16.02.2021 17:40

Mathematics, 16.02.2021 17:50



