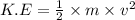Chemistry, 30.07.2019 00:00 querty2400
In a chemical reaction occurring in an open beaker, the reactants have a mass of 10.0 g and contain 1000 j of energy. the products in the beaker contain a mass of 5.0 g and 500 j of energy. which best explains the difference in energy and mass as reactants are converted to products? kinetic energy closed systems law of conservation electromagnetic energy

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:00, daytonalive83481
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 16:50, TheOriginal2x
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3
You know the right answer?
In a chemical reaction occurring in an open beaker, the reactants have a mass of 10.0 g and contain...
Questions in other subjects:



Biology, 26.03.2020 22:54

Biology, 26.03.2020 22:54

Advanced Placement (AP), 26.03.2020 22:54

Mathematics, 26.03.2020 22:54

History, 26.03.2020 22:54

History, 26.03.2020 22:54

Mathematics, 26.03.2020 22:54

Mathematics, 26.03.2020 22:54