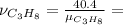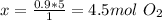The combustion of propane may be described by the chemical equation,
c3h8(g) + 5o2(g) -->...

Chemistry, 21.08.2019 12:00 natachalebrun2
The combustion of propane may be described by the chemical equation,
c3h8(g) + 5o2(g) --> 3co2(g) + 4h2o(g)
how many grams of o2(g) are needed to completely burn 40.4 g of c3h8(g)?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:20, mgavyn1052
Calculate the molarity of the solution. 6.02 x 1022 molecules of hci (molecular weight = 36.5 g/mole) in 2.0 liters of water m
Answers: 1

Chemistry, 22.06.2019 00:30, mannster03
This active feature of earth's crust in building mountain ranges as well as islands. this feature is a a) cavern. b) earthquake. c) mountain. d) volcano.
Answers: 2

Chemistry, 22.06.2019 07:30, isalih7256
Free answer. the treaty of versailles ended world war i, but some of the terms of the treaty contributed to the beginning of world war ii. which was one of the terms of the treaty? the answer would be "germany was forces to pay reparations to the allied countries.". i hope this .
Answers: 1

Chemistry, 22.06.2019 12:40, whitethunder05
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2
You know the right answer?
Questions in other subjects:



Mathematics, 14.09.2021 17:10

Mathematics, 14.09.2021 17:10



Biology, 14.09.2021 17:10

Mathematics, 14.09.2021 17:10


Mathematics, 14.09.2021 17:10