
Chemistry, 02.08.2019 02:00 daydallas01
Carbon has four valence electrons, and oxygen has six valence electrons. if carbon and oxygen bond covalently, which of the following is the correct lewis dot (electron dot) structure for carbon dioxide?
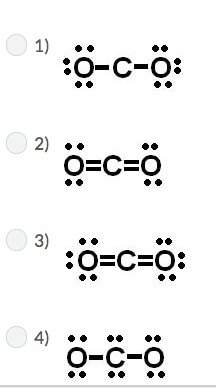

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, juansantos7b
Which of the following statements is true about planck’s law
Answers: 1

Chemistry, 22.06.2019 00:30, megaaan214p61pb7
Which compounds have the empirical formula ch2o? a. c2h4o2 b. c3h6o3 c. ch2o2 d. c5h10o5 e. c6h12o6
Answers: 3

Chemistry, 22.06.2019 04:30, anthony4034
Use the periodic table to determine the electron configuration of dysprosium (dy) and americium (am) in noble-gas notation.
Answers: 1

Chemistry, 22.06.2019 04:40, khan2491
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2
You know the right answer?
Carbon has four valence electrons, and oxygen has six valence electrons. if carbon and oxygen bond c...
Questions in other subjects:

Computers and Technology, 28.06.2019 13:00


Mathematics, 28.06.2019 13:00


Mathematics, 28.06.2019 13:00


Social Studies, 28.06.2019 13:00


Social Studies, 28.06.2019 13:00



