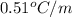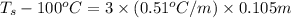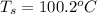Chemistry, 02.08.2019 08:00 jojo492192
Calculate the freezing point of the solution containing 0.105 m k2s. calculate the boiling point of the solution above. -g

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:10, yootmytoot
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution. calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

You know the right answer?
Calculate the freezing point of the solution containing 0.105 m k2s. calculate the boiling point of...
Questions in other subjects:

Mathematics, 25.08.2020 20:01





Advanced Placement (AP), 25.08.2020 20:01

Mathematics, 25.08.2020 20:01

English, 25.08.2020 20:01


Mathematics, 25.08.2020 20:01

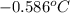
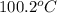
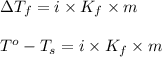
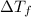 = change in freezing point
= change in freezing point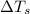 = freezing point of solution = ?
= freezing point of solution = ?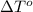 = freezing point of water =
= freezing point of water = 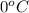
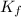 = freezing point constant for water =
= freezing point constant for water = 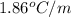
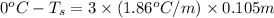
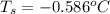
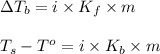
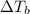 = change in boiling point
= change in boiling point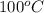
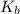 = boiling point constant for water =
= boiling point constant for water =