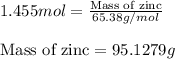Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:40, CylieTbh
Which statement is usually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 3


Chemistry, 22.06.2019 08:00, PrincessKeliah5538
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1
You know the right answer?
Nitric acid and zinc react to form zinc nitrate, ammonium nitrate, and water. how many atoms of zinc...
Questions in other subjects:

Spanish, 15.07.2019 13:00

History, 15.07.2019 13:00



Spanish, 15.07.2019 13:00

Arts, 15.07.2019 13:00




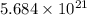 atoms.
atoms.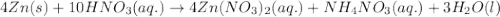
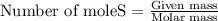 ....(1)
....(1)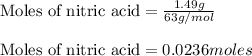
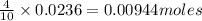 of zinc metal
of zinc metal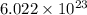 number of atoms.
number of atoms.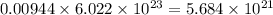 number of atoms.
number of atoms.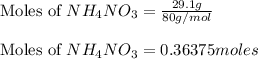
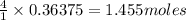 of zinc.
of zinc.