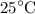Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:00, natalie857123
For each of the following types of reactions, write a general reaction formula in the symbolic form—for example, a + b → ab. single-displacement double-displacement synthesis decomposition
Answers: 1


Chemistry, 22.06.2019 14:30, clemsongirl5392
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2

Chemistry, 22.06.2019 20:00, kalcius9698
What is the molarity of the solution produced when 145 g of nacl is dissolved in sufficient water to prepare 2.75 l of solution?
Answers: 1
You know the right answer?
Dimethylamine, (ch3)2nh, has a kb value of 5.4 x 10â4. in a 1.2 m solution of dimethylamine at 25ëc...
Questions in other subjects:


Mathematics, 21.09.2019 09:30

Social Studies, 21.09.2019 09:30


English, 21.09.2019 09:30

Social Studies, 21.09.2019 09:30

Mathematics, 21.09.2019 09:30


English, 21.09.2019 09:30

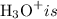
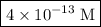
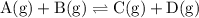
![{\text{K}}=\dfrac{{\left[ {\text{D}}\right]\left[{\text{C}}\right]}}{{\left[{\text{A}} \right]\left[{\text{B}}\right]}}](/tpl/images/0162/8999/3958d.png)
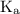 and equilibrium constant for the dissociation of base is known as
and equilibrium constant for the dissociation of base is known as 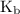 .
.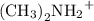 and hydroxide ion.
and hydroxide ion.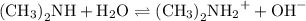
![{{\text{K}}_b}=\dfrac{{\left[{{{\left({{\text{C}}{{\text{H}}_3}}\right)}_2}{\text{N}}{{\text{H}}_2}^+} \right]\left[\\{{\text{O}}{{\text{H}}^-}}\right]}}{{\left[{{{\left({{\text{C}}{{\text{H}}_3}}\right)}_2}{\text{NH}}}\right]}}](/tpl/images/0162/8999/2ee99.png) ...... (1)
...... (1)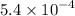 .
.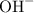 becomes x at equilibrium.
becomes x at equilibrium.![\left[ {{\text{O}}{{\text{H}}^ - }}\right]](/tpl/images/0162/8999/30776.png) , x for
, x for ![\left[ {{{\left( {{\text{C}}{{\text{H}}_3}} \right)}_2}{\text{N}}{{\text{H}}_2}^ + } \right]](/tpl/images/0162/8999/8fe93.png) and 1.2-x for
and 1.2-x for ![\left[{{{\left({{\text{C}}{{\text{H}}_3}}\right)}_2}{\text{NH}}} \right]](/tpl/images/0162/8999/70fe2.png) in equation (1).
in equation (1).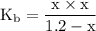 ...... (2)
...... (2) 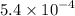 for
for 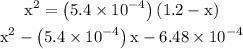
![\left[{{\text{O}}{{\text{H}}^ - }}\right]](/tpl/images/0162/8999/91da6.png) of is equal to x and therefore the concentration of
of is equal to x and therefore the concentration of ![\left[{{{\text{H}}_{\text{3}}}{{\text{O}}^ + }}\right]\left[{{\text{O}}{{\text{H}}^ - }}\right] = {10^{ - 14}}](/tpl/images/0162/8999/199d1.png) ...... (3)
...... (3)![\left[{{{\text{H}}_3}{{\text{O}}^ + }}\right]](/tpl/images/0162/8999/496c8.png) is the concentration of hydronium ion.
is the concentration of hydronium ion.![\left[ {{{\text{H}}_{\text{3}}}{{\text{O}}^ + }} \right]=\dfrac{{{{10}^{ - 14}}}}{{\left[ {\text{O}}{{\text{H}}^ - }}\right]}}](/tpl/images/0162/8999/e7aaf.png) …… (4)
…… (4) ![\begin{aligned}\left[{{{\text{H}}_{\text{3}}}{{\text{O}}^ + }}\right]&=\frac{{{{10}^{ - 14}}}}{{0.02572}}\\&= 3.8880\times{10^{ - 13}}\;{\text{M}}\\&=4\times {10^{ - 13}}\;{\text{M}}\\\end{aligned}](/tpl/images/0162/8999/9343b.png)