Me i beg you
the average kinetic energy of 1 mole of a gas at -32 degrees celsius is: <...

Me i beg you
the average kinetic energy of 1 mole of a gas at -32 degrees celsius is:
3.01 x 103 j
3.99 x 102 j
-3.99 x 102 j
3.80 x 103 j
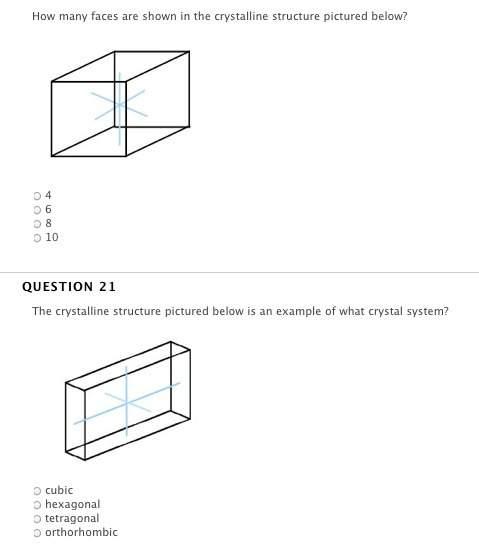
the relationship between volume and temperature of a gas, when pressure and moles of a gas are held constant, is: v*t = k.
true
false
the relationship between moles and volume, when pressure and temperature of a gas are held constant, is: v/n = k. we could say then, that:
if the moles of gas are tripled, the volume must also triple.
if the moles of gas are halved, the volume must be doubled.
if the moles of gas are doubled, the volume must be halved.
if the moles of gas are doubled, the volume is unaffected.
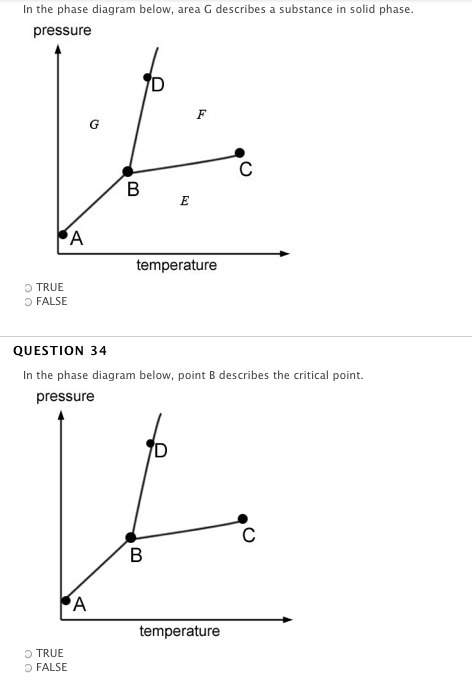
if the temperature and volume of a gas are held constant, an increase in pressure would most likely be caused by an increase in the number of moles of gas.
true
false
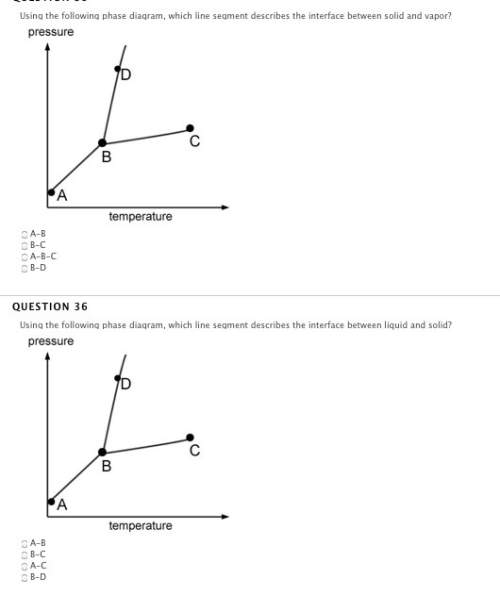
if the vapor pressure of a liquid is less than the atmospheric pressure, the liquid will not boil.
true
false


Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:00, hemolelekeakua
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 04:00, lucasrandall
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution?
Answers: 2

Chemistry, 22.06.2019 14:30, cxttiemsp021
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3

You know the right answer?
Questions in other subjects:




Mathematics, 10.07.2019 00:30


Mathematics, 10.07.2019 00:30


History, 10.07.2019 00:30




