
Chemistry, 03.08.2019 13:30 GreenHerbz206
Anitrogen oxide, containing 53.85% n, acts as a vasodilator, lowering blood pressure in the human body. what is its empirical formula? '

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 22:30, britotellerialuis
Ibeg i need 20. a reaction produces 4.93 l of oxygen, but was supposed to produce 1 mol of oxygen. what is the percent yield?
Answers: 1

Chemistry, 22.06.2019 03:30, asianaenaeh
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 05:10, hadellolo8839
How many miles of water are produced if 5.43 mol pbo2 are consumed
Answers: 1
You know the right answer?
Anitrogen oxide, containing 53.85% n, acts as a vasodilator, lowering blood pressure in the human bo...
Questions in other subjects:



Engineering, 12.03.2020 06:34


Mathematics, 12.03.2020 06:34

Biology, 12.03.2020 06:34

Mathematics, 12.03.2020 06:34

Mathematics, 12.03.2020 06:34


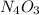 . Empirical formula is the smallest whole number ratios of elements in a compound.
. Empirical formula is the smallest whole number ratios of elements in a compound. 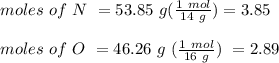
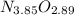
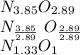
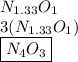
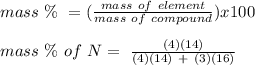
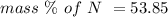
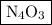 .
.
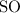 .
.
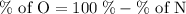 …… (1)
…… (1)
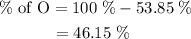
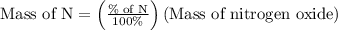 …… (2)
…… (2)
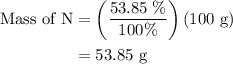
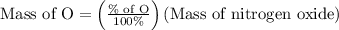 …… (3)
…… (3)
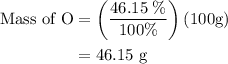
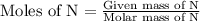 …… (5)
…… (5)
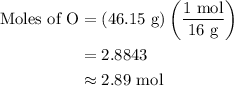
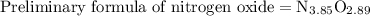
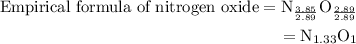

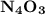 .
.


