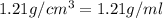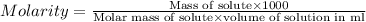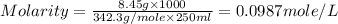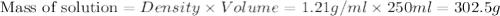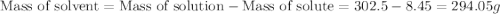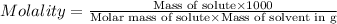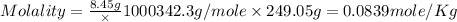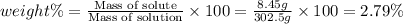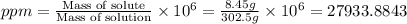Chemistry, 04.08.2019 17:30 fredvales19
Asolution of a sugar with the chemical formula (c12h22o11) is prepared by dissolving 8.45 g in 250.0 ml of water at 25 c. the density of this solution is 1.21 g/cm3. calculate the concentration in terms of molarity, molality, weight percent and ppm. assume that the volume of the solution is equal to the volume of the solvent. show your work.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, drivinghydra
What is the relation between concentration of reactants and the rate of chemical reaction?
Answers: 1


Chemistry, 22.06.2019 21:30, steven0448
An atomic nucleus is composed ofa)protons. b)protons and neutrons. c)protons and electrons. d)protons, neutrons, and electrons.
Answers: 1
You know the right answer?
Asolution of a sugar with the chemical formula (c12h22o11) is prepared by dissolving 8.45 g in 250.0...
Questions in other subjects:







Computers and Technology, 31.08.2020 01:01



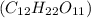 = 8.45 g
= 8.45 g