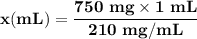Chemistry, 05.08.2019 00:30 minnie7760
Calculate how much 95% ethyl alcohol will be required to dissolve 0.75 g of sulfanilamide at 78°c

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, lindseyklewis1p56uvi
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1


Chemistry, 22.06.2019 11:30, chelseychew32
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2

Chemistry, 22.06.2019 13:10, bartonamber4042
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1
You know the right answer?
Calculate how much 95% ethyl alcohol will be required to dissolve 0.75 g of sulfanilamide at 78°c...
Questions in other subjects:


Social Studies, 15.10.2019 22:40




Biology, 15.10.2019 22:40



Social Studies, 15.10.2019 22:40

Mathematics, 15.10.2019 22:40