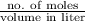Chemistry, 05.08.2019 01:50 harmonyfern5648
How would a solution that is labeled 5.0 m would be read as? 5.0 molar 5.0 moles 5.0 concentrated 5.0 dilute

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 12:10, kaitlynbernatz2778
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3
You know the right answer?
How would a solution that is labeled 5.0 m would be read as? 5.0 molar 5.0 moles 5.0 concentrated 5...
Questions in other subjects:


English, 02.12.2020 16:20