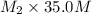Chemistry, 01.08.2019 02:10 nxusasmangaliso1191
If you have 25.0 ml of a 6.50m naoh solution and you add 10.0 ml of water to it, what is the new concentration?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:10, josephpezza18
The covalent compound acetylene, which is the fuel of the oxyacetylene torch used by welders, has the molecular formula c2h2. the covalent compound benzene, a commercial solvent, has the molecular formula c6h6 each of these covalent compounds contains carbon and hydrogen atoms in a one-to-one ratio. would it be correct to write the chemical formulas of each as ch? explain.
Answers: 1

Chemistry, 22.06.2019 11:30, ashleybarrera2000
For each of the following compounds, decide whether the compound's solubility in aqueous solution changes with ph. if the solubility does change, pick the ph at which you'd expect the highest solubility. you'll find ksp data in the aleks data tab. compounds does solubility change with ph
Answers: 3

Chemistry, 22.06.2019 12:30, americanbellabeauty
Acontrol during an experiment. might change remains constant does not exist does change
Answers: 1
You know the right answer?
If you have 25.0 ml of a 6.50m naoh solution and you add 10.0 ml of water to it, what is the new con...
Questions in other subjects:

Mathematics, 22.07.2019 12:30

Biology, 22.07.2019 12:30




Health, 22.07.2019 12:30

English, 22.07.2019 12:30



World Languages, 22.07.2019 12:30

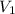 = 25.0 ml,
= 25.0 ml, 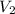 = (10.0 ml + 25.0 ml) = 35.0 ml
= (10.0 ml + 25.0 ml) = 35.0 ml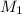 = 6.50 M,
= 6.50 M, 
 =
= 
 =
=