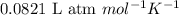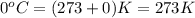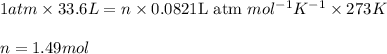Chemistry, 26.07.2019 06:50 mazolethrin3461
How many moles of oxygen (o2) are present in 33.6 l of the gas at 1 atm and 0°c?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 23.06.2019 02:30, ggpro4life3000
Ascientist wants to know how individual lions within a pride interact with each other in their own environment. to do this, the scientist sedates and tags all of the lions within a pride. then, he places several remotely-controlled video cameras near the lions' den and performs an observational field study. he collects continuous video footage over the span of one year, analyzes the video, and then forms conclusions based on his observations.
Answers: 2

Chemistry, 23.06.2019 07:00, MathChic68
Why do the strengths of london (dispersion) forces generally increase with increasing molecular size? choose one: a. heavier atoms have stronger attractions for each other than lighter atoms. b. dispersion forces are all equal in magnitude; there is no size dependence. c. dispersion forces arise from the attraction between the nuclei of atoms, and larger molecules have larger nuclei. d. dispersion forces arise from dipoles caused by the electron distribution being distorted. larger molecules have more electrons and, therefore, more distortions and a bigger force. e. dispersion forces depend on distance. larger molecules are farther apart and so the forces are smaller.
Answers: 2

Chemistry, 23.06.2019 11:30, kayabwaller4589
A) equal lines b) parallel lines c) perpendicular lines d) none of the above
Answers: 1
You know the right answer?
How many moles of oxygen (o2) are present in 33.6 l of the gas at 1 atm and 0°c?...
Questions in other subjects:


Biology, 12.10.2019 10:00

Geography, 12.10.2019 10:00




Mathematics, 12.10.2019 10:00

Social Studies, 12.10.2019 10:00

Mathematics, 12.10.2019 10:00

English, 12.10.2019 10:00