Which missing item would complete this beta decay reaction?
...

Chemistry, 27.09.2019 19:40 Jerryholloway5871
Which missing item would complete this beta decay reaction?
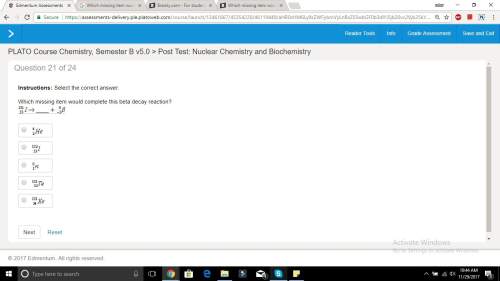

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 10:20, blondielocks2002
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3
You know the right answer?
Questions in other subjects:






Biology, 09.10.2019 19:50

English, 09.10.2019 19:50



Chemistry, 09.10.2019 19:50



