
Chemistry, 23.07.2019 20:00 bobbyxii6033
Hydrogen sulfate reacts with water to produce sulfate ions and hydronium ions: hso4−+h2o⇌so42−+h3o+ identify the conjugate acid-base pairs.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, alexandroperez13
It takes 945.kj/mol to break a nitrogen-nitrogen triple bond. calculate the maximum wavelength of light for which a nitrogen-nitrogen triple bond could be broken by absorbing a single photon.
Answers: 3

Chemistry, 21.06.2019 23:30, hellokitty1647
For the following dehydrohalogenation (e2) reaction, draw the zaitsev product(s) resulting from elimination involving c3–c4 (i. e., the carbon atoms depicted with stereobonds). show the product stereochemistry clearly. if there is more than one organic product, both products may be drawn in the same box. ignore elimination involving c3 or c4 and any carbon atom other than c4 or c3.
Answers: 3

Chemistry, 22.06.2019 03:50, sgslayerkingminecraf
Which of the following statements about acidic water is true? a. acid has no effect on the h, o molecules. b. the solution contains a larger number of oh ions than h, o ions. c. the solution contains a larger number of h, o ions than qh ions. d. the solution contains an equal number of h, o ions and oh ions. none of the above e.
Answers: 1
You know the right answer?
Hydrogen sulfate reacts with water to produce sulfate ions and hydronium ions: hso4−+h2o⇌so42−+h3o+...
Questions in other subjects:

English, 22.07.2020 05:01

Mathematics, 22.07.2020 05:01



Mathematics, 22.07.2020 05:01

Mathematics, 22.07.2020 05:01

Mathematics, 22.07.2020 05:01

History, 22.07.2020 05:01


Mathematics, 22.07.2020 05:01


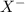 ), and the conjugate acid-base pair is
), and the conjugate acid-base pair is 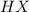 -
- 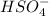 ) is an ion from sulfuric acid. It is still an acid in itself and can "react" with water ((
) is an ion from sulfuric acid. It is still an acid in itself and can "react" with water ((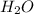 ) to form the sulfate (
) to form the sulfate (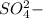 ) and hydronium (
) and hydronium (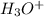 )ions.
)ions. 
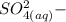 is identified to be the conjugate of the acid
is identified to be the conjugate of the acid 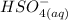 . Thus, the conjugate acid-base pair is
. Thus, the conjugate acid-base pair is 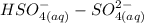 .
.


