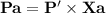Chemistry, 22.07.2019 11:00 mercedesamatap21hx0
Three gases (8.00 g of methane, ch4, 18.0 g of ethane, c2h6, and an unknown amount of propane, c3h8) were added to the same 10.0-l container. at 23.0 ∘c, the total pressure in the container is 5.50 atm . calculate the partial pressure of each gas in the container.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, vlactawhalm29
Select the correct text in the passage. which sentences describe examples of sustainable living? i live in an old apartment building downtown, but my company is based in an office park on the outskirts of the city. i drive an old car that needs to be replaced. i plan to buy a hybrid for better gas mileage, but for now i am able to carpool with a couple of friends from work. the drive to the office park is about 45 minutes each way, but we do get to work in a modern building. the architects just received a leed certification for the design.
Answers: 3

Chemistry, 22.06.2019 08:30, ebigham5117
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 2

Chemistry, 22.06.2019 12:30, kingbot350
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1
You know the right answer?
Three gases (8.00 g of methane, ch4, 18.0 g of ethane, c2h6, and an unknown amount of propane, c3h8)...
Questions in other subjects:

Physics, 03.02.2022 14:00





SAT, 03.02.2022 14:00




Mathematics, 03.02.2022 14:00