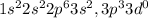Chemistry, 15.07.2019 15:10 angryapple1133
Based on its location in the periodic table, what can most likely be predicted about phosphorus? a. it has three valence electrons available for bonding. b. it has three nonbonding electrons. c. it has five valence electrons available for bonding. d. it has five nonbonding electrons.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, advancedgamin8458
Consider the nuclear equation below. 239 > x + 4 he 94 2 what is x? 1.235 cm 96 2.243 u 92 3.235 u 92 4.243 cm 96
Answers: 2


Chemistry, 22.06.2019 19:30, simihehe
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2

Chemistry, 22.06.2019 23:00, jolainjoseph01998
What element has similar physical and chemical properties as boron.
Answers: 1
You know the right answer?
Based on its location in the periodic table, what can most likely be predicted about phosphorus? a....
Questions in other subjects:


Arts, 12.05.2021 19:10



English, 12.05.2021 19:10


Mathematics, 12.05.2021 19:10

Biology, 12.05.2021 19:10


English, 12.05.2021 19:10