Chemistry, 13.07.2019 07:30 orlandocruz42
If a system has 425 kcal of work done to it, and releases 5.00 × 102 kj of heat into its surroundings, what is the change in internal energy of the system?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, logan12345677885675
What are the primary responsibilities of a chemical engineer involved in "r& d"? develop large scale manufacturing operations discover new products and processes training of new chemists determine products needed by consumers
Answers: 2


Chemistry, 22.06.2019 20:20, carcon2019
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 23.06.2019 01:00, jazzy200232
Which process results in the release of energy stored in the products of photosynthesis? a. polymer synthesis b. depolymerization c. digestion d. cellular respiration
Answers: 1
You know the right answer?
If a system has 425 kcal of work done to it, and releases 5.00 × 102 kj of heat into its surrounding...
Questions in other subjects:



Mathematics, 20.10.2020 18:01

Mathematics, 20.10.2020 18:01





History, 20.10.2020 18:01

Biology, 20.10.2020 18:01

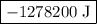 .
.
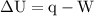 …… (1)
…… (1) 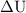 is the change in internal energy of the system.
is the change in internal energy of the system.
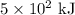 .
.