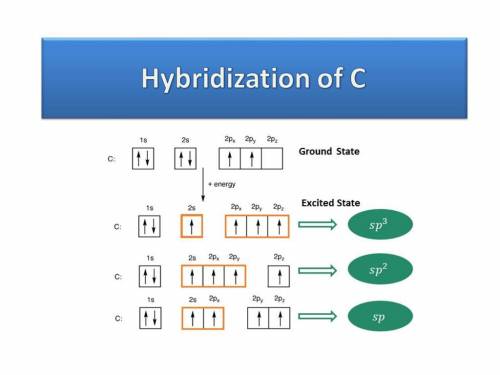
Chemistry, 17.09.2019 18:00 anjumuddin9
What orbitals are used to form each bond in methanol, ch3oh? be sure to answer all parts?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, erikloza12pdidtx
Which type of bond is present in hydrogen sulfide (h2s)? the table of electronegativities is given. a. hydrogen b. ionic c. nonpolar covalent d. polar covalent
Answers: 1

Chemistry, 22.06.2019 23:00, DESI111609
What is the average rate of the reaction between 10 and 20 s?
Answers: 1


You know the right answer?
What orbitals are used to form each bond in methanol, ch3oh? be sure to answer all parts?...
Questions in other subjects:


Geography, 26.10.2020 07:30






English, 26.10.2020 07:30

Mathematics, 26.10.2020 07:30