Calculate the molarity of 6.631 g NANO3 in 100.0 mL of solution
add stepss...

Chemistry, 01.05.2022 21:50 glstephens04
Calculate the molarity of 6.631 g NANO3 in 100.0 mL of solution
add stepss

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:00, happy121906
Which position represents spring in the southern hemisphere? a) b) c) d)
Answers: 2

Chemistry, 22.06.2019 08:00, gomezyonathan93
An observation that requires measurement is called quantitative observable or qualitative
Answers: 1


Chemistry, 22.06.2019 10:40, yfgkeyonna
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2
You know the right answer?
Questions in other subjects:




Mathematics, 11.03.2020 06:15


Mathematics, 11.03.2020 06:15



Mathematics, 11.03.2020 06:16

Mathematics, 11.03.2020 06:16

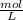
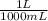 = 0.1000 L
= 0.1000 L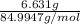 = 0.07802 mol
= 0.07802 mol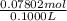 = 0.7802 M
= 0.7802 M

