
Chemistry, 16.03.2022 20:00 kierafisher05
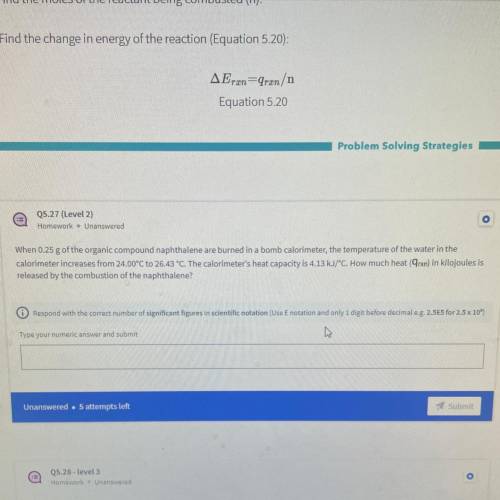
When 0.25 g of the organic compound naphthalene are burned in a bomb calorimeter, the temperature of the water in the
calorimeter increases from 24.00°C to 26.43 °C. The calorimeter's heat capacity is 4.13 kJ/°C. How much heat (9rxn) in kilojoules is
released by the combustion of the naphthalene?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:00, kangasc6124
Which type of orbits are found in the principal energy level n = 2 a - s b - s, f c - s, d d - s, p e - s, p, d
Answers: 1

Chemistry, 22.06.2019 06:00, lindseyklewis1p56uvi
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 10:10, andersonemma2222
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1
You know the right answer?
When 0.25 g of the organic compound naphthalene are burned in a bomb calorimeter, the temperature of...
Questions in other subjects:

Mathematics, 25.11.2021 21:20

History, 25.11.2021 21:20


Mathematics, 25.11.2021 21:20

Mathematics, 25.11.2021 21:20

English, 25.11.2021 21:20



English, 25.11.2021 21:20

Chemistry, 25.11.2021 21:20



