
Chemistry, 14.02.2022 16:50 stupidsmoke4272
HERE
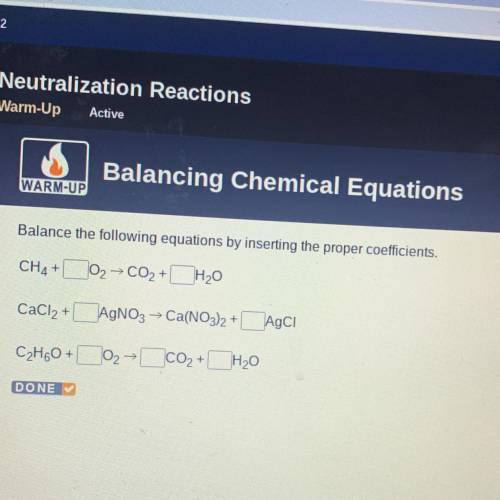
Balance the following equations by inserting the proper coefficients.
CH4 +
102 → CO2 + H20
CaCl2 +
AgNO3 → Ca(NO3)2 +Agci
C2H60+
+
02
co2+ H20

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 18:30, rosie20052019
You open a can of soda at room temperature and hear a hiss. which of the following factors has changed inside the container? a.) atmospheric pressure b.) temperature of gas c.) type of gas d.) amount of gas
Answers: 1

Chemistry, 22.06.2019 22:30, robertss403
How many moles of kci are produced from 2.50 moles k
Answers: 1

Chemistry, 23.06.2019 07:30, shealynh52
Achemist at a pharmaceutical company is measuring equilibrium constants for reactions in which drug candidate molecules bind to a protein involved in cancer. the drug molecules bind the protein in a 1: 1 ratio to form a drug-protein complex. the protein concentration in aqueous solution at 25 ˚c is 1.74 x10-6 m . drug a is introduced into the protein solution at an initial concentration of 2.00 x10-6m. drug b is introduced into a separate, identical protein solution at an initial concentration of 2.00 x10-6m. at equilibrium, the drug a-protein solution has an a-protein complex concentration of 1.00 x10-6m, and the drug b solution has a b-protein complex concentration of 1.40 x10-6m. a. calculate the kc value for the a-protein binding reaction. b. calculate the kc value for the b-protein binding reaction. c. assuming that the drug that binds more strongly will be more effective, which drug is the better choice for further research?
Answers: 1
You know the right answer?
HERE
Balance the following equations by inserting the proper coefficients.
CH4 +
102 →...
CH4 +
102 →...
Questions in other subjects:


Mathematics, 30.11.2020 23:50

Mathematics, 30.11.2020 23:50

Mathematics, 30.11.2020 23:50



Physics, 30.11.2020 23:50

English, 30.11.2020 23:50

Mathematics, 30.11.2020 23:50

History, 30.11.2020 23:50



