
Chemistry, 14.02.2022 09:00 whereswoodruff
The following molecular equation represents the reaction that occurs when aqueous solutions of silver(I) nitrate and barium chloride are combined.
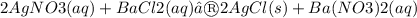
Write the balanced net ionic equation for the reaction.
(Use the lowest possible coefficients. Be sure to specify states such as (aq) or (s). If a box is not needed, leave it blank.)

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:20, ayoismeisalex
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1



Chemistry, 23.06.2019 00:40, joe7977
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1
You know the right answer?
The following molecular equation represents the reaction that occurs when aqueous solutions of silve...
Questions in other subjects:



Mathematics, 10.06.2020 09:57


Mathematics, 10.06.2020 09:57

Mathematics, 10.06.2020 09:57

Health, 10.06.2020 09:57


Social Studies, 10.06.2020 09:57




