
Chemistry, 13.02.2022 04:40 DesireeGuzman5213
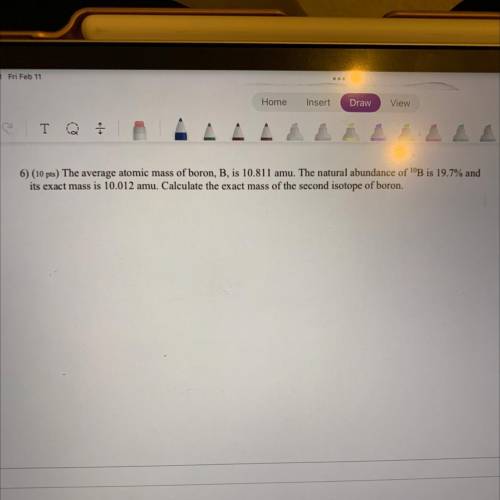
6) The average atomic mass of boron, B, is 10.811 amu. The natural abundance of 10B is 19.7% and it’s exact mass is 10.012 amu. Calculate the exact mass of the second isotope of boron.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, Blaise2653
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1

Chemistry, 22.06.2019 23:00, maddyleighanne
Arectangle has a diagonal 20 inches long that forms angles of 60 and 30 with the sides. find the perimeter of the rectangle. for geometry
Answers: 3

You know the right answer?
6) The average atomic mass of boron, B, is 10.811 amu. The natural abundance of 10B is 19.7% and it’...
Questions in other subjects:


Mathematics, 15.07.2019 02:30





Mathematics, 15.07.2019 02:30

Mathematics, 15.07.2019 02:30




