Given the equation
N2(g) +3H2 (g)--> 2NH3 (g)
1 mole of N2 gas is needed to completely re...

Chemistry, 25.12.2021 17:40 levicorey846
Given the equation
N2(g) +3H2 (g)--> 2NH3 (g)
1 mole of N2 gas is needed to completely react with 3 moles of H2 gas.
How many molecules of H2 gas are needed?


Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:50, fhemmie963
What type of reaction is illustrated? 2c12o5 = 2cl2 + 502
Answers: 2

Chemistry, 22.06.2019 14:00, Killion2022
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1

Chemistry, 22.06.2019 15:00, makaylajones74pdxtrk
What is the most important factor in determining climates.
Answers: 1

Chemistry, 22.06.2019 22:00, notearslefttocry14
Imagine one batch of soup (batch “a”) is made with 8.19 g/can of salt, according to the recipe, and a second batch of soup (batch “b”) is made with 8.32 g/can of salt. explain which batch would be more resistant to frost damage if it is shipped a great distance in winter and explain why.
Answers: 2
You know the right answer?
Questions in other subjects:

Social Studies, 18.12.2021 05:30

History, 18.12.2021 05:30

Social Studies, 18.12.2021 05:30



Arts, 18.12.2021 05:30



Physics, 18.12.2021 05:30

 molecules H2
molecules H2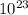 molecules= 1.81 x
molecules= 1.81 x 

