
Chemistry, 15.12.2021 23:20 juelchasse
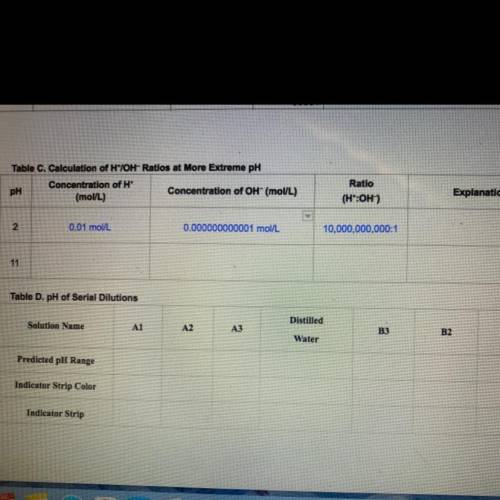
In Step 5, you will calculate H*/OH- ratios for more
extreme pH solutions. Find the concentration of H* ions
to OH- ions listed in Table B of your Student Guide for a
solution at a pH = 2. Then divide the H* concentration by
the OH concentration. Record these concentrations and
ratio in Table C.
I know the answer I just don’t know what to put for the explanation on the table.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, amandasantiago2001
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3

Chemistry, 22.06.2019 06:00, palomaresmitchelle
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 18:30, sarahbug56
Which rate indicates the number of children that would be born per woman if she were to live to the end of her child bearing years
Answers: 2

Chemistry, 22.06.2019 20:30, ShahinF7536
Some familiar products contain some of the same types of atoms. for instance, the chemical formula for baking soda is nahco 3. the chemical formula for liquid bleach is naclo, and the chemical formula for table salt is nacl. which choice best describes why these three products have some of the same types of atoms in common?
Answers: 1
You know the right answer?
In Step 5, you will calculate H*/OH- ratios for more
extreme pH solutions. Find the concentration...
Questions in other subjects:

History, 28.05.2021 09:50

English, 28.05.2021 09:50



Mathematics, 28.05.2021 09:50



Biology, 28.05.2021 09:50

Engineering, 28.05.2021 09:50



