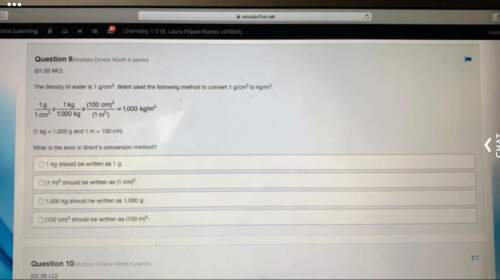
2 g (an excess) of iron is added to 50 cm3 of 0.5 M sulfuric acid. When the reaction is over, the reaction mixture is filtered. The mass of the unreacted iron is found to be 0.6 g. (Ar: Fe = 56.)
a. What mass of iron took part in the reaction?
b. How many moles of iron atoms took part?
c. How many moles of sulfuric acid reacted?
d. Write the equation for the reaction, and deduce the charge on the iron ion that formed.
e. What volume of hydrogen (calculated at itp) bubbled off during the reaction?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, jamesnaquan132
What is the most stable monatomic ion formed from nitrogen
Answers: 2

Chemistry, 22.06.2019 15:00, levelebeasley1
Which theory was contradicted by experiments with the photoelectric effect? light spreads out after it passes through a small opening. as soon as light strikes metal, electrons will be ejected. visible light, regardless of color, will cause the ejection of electrons when striking metal. the kinetic energy of ejected electrons depends on the frequency of light that strikes the metal.
Answers: 2

You know the right answer?
2 g (an excess) of iron is added to 50 cm3 of 0.5 M sulfuric acid. When the reaction is over, the re...
Questions in other subjects:

Biology, 14.01.2020 22:31



History, 14.01.2020 22:31



Health, 14.01.2020 22:31

Mathematics, 14.01.2020 22:31

Mathematics, 14.01.2020 22:31

Mathematics, 14.01.2020 22:31