Chemistry, 06.12.2021 21:20 ericchen4399
A mass of 1.01 g carbon monoxide is reacted with 5.95 g hydrogen to form methanol.
CO(g) + 2 H2(g) → CH3OH(g)
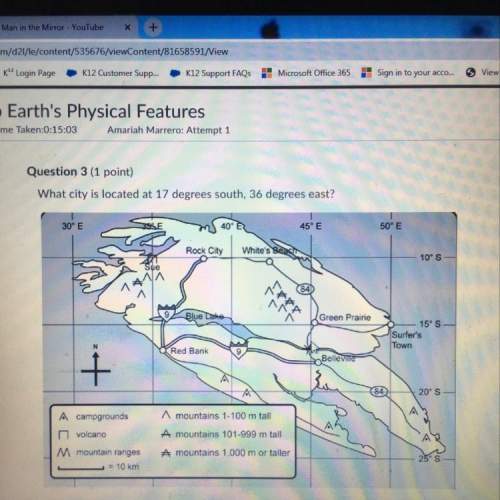
The reaction is performed in a 5.00 L flask at 85.0 °C and proceeds to completion. What is the partial pressure (in mmHg) of each of the three species following completion of the reaction? What is the total pressure in the flask?
CO ? mmHg
H2 ? mmHg
CH3OH ? mmHg
Total ? mmHg

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, poopybutt541
Avariable that is not being directly tested during an experiment should be
Answers: 1

Chemistry, 23.06.2019 01:20, cedricevans41p4j3kx
Use the de broglie's wave equation to find the wavelength of an electron moving at 4.5 × 106 m/s. show your work. note: h= plank's constant (6.62607 x 10-34 j s)
Answers: 1

You know the right answer?
A mass of 1.01 g carbon monoxide is reacted with 5.95 g hydrogen to form methanol.
CO(g) + 2 H2(g)...
Questions in other subjects:



Mathematics, 25.11.2019 19:31







Chemistry, 25.11.2019 19:31