Chemistry, 05.12.2021 14:00 idontknow1993
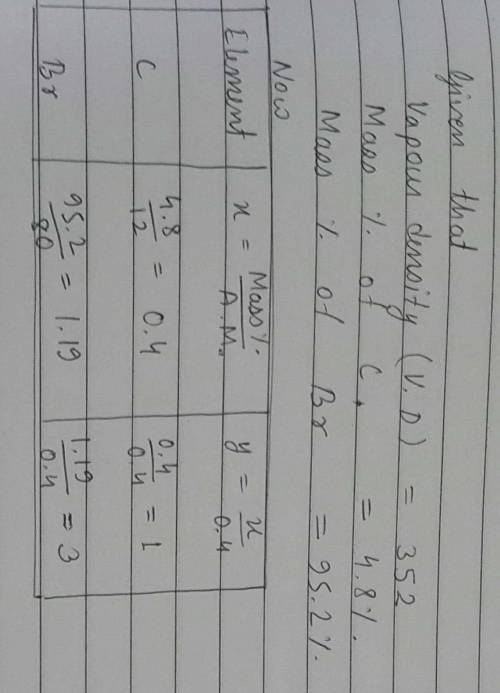
2. A compound with an empirical formula of C2H3Br2 has a molar mass of 373.69 g/mol. What is
the molecular formula?
(The molar mass of C2H3Br2 = 186.85 g/mol)
E) C4H6Br4
А) C2H3Br2
B) CHBr
C) C6H9Br6 D) C4H6Br2
(0.5 Points)
A

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, alaina3792
Of the groups of elements below, which are most likely to gain electrons to become anions? a. alkali metal b. boron group c. halogen d. transition metal
Answers: 2


Chemistry, 23.06.2019 10:30, lvoltin1073
When a chemist collects hydrogen gas over water, she ends up with a mixture of hydrogen and water vapor in her collecting bottle if the pressure in the collecting bottle is 97.1 kilopascals and the vapor pressure of the water is 3 2 kilopascals, what is the partial pressure of the hydrogen?
Answers: 1

Chemistry, 23.06.2019 11:30, ssuereichard
If this sedimentary rock layer is truly the oldest one of marine origin, what do you think that tells usabout the formation of earth's oceans?
Answers: 2
You know the right answer?
2. A compound with an empirical formula of C2H3Br2 has a molar mass of 373.69 g/mol. What is
the m...
Questions in other subjects:




English, 13.01.2021 01:40

Mathematics, 13.01.2021 01:40

History, 13.01.2021 01:40

Biology, 13.01.2021 01:40

Chemistry, 13.01.2021 01:40

Mathematics, 13.01.2021 01:40