
Chemistry, 01.12.2021 20:20 agarcia24101993
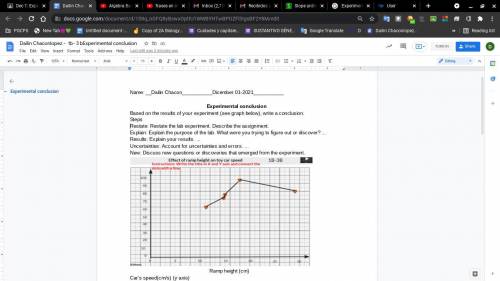
Based on the results of your experiment (see graph below), write a conclusion.
Steps
Restate: Restate the lab experiment. Describe the assignment.
Explain: Explain the purpose of the lab. What were you trying to figure out or discover? ...
Results: Explain your results. ...
Uncertainties: Account for uncertainties and errors. ...
New: Discuss new questions or discoveries that emerged from the experiment.
write a conclision.. Please HELP

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 23:00, poolwaterisgross
How does the value of the equilibrium constant show that a reaction reaches equilibrium very quickly? (a) the equilibrium constant is large. (b) the equilibrium constant is small. (c) the equilibrium constant is zero. (d) the value of the equilibrium constant does not show how quickly a reaction comes to equilibrium.
Answers: 1

Chemistry, 23.06.2019 00:20, HernanJe6
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1


Chemistry, 23.06.2019 14:00, lilyella1004
How does electronegativity changes as we move from left to right across a period
Answers: 2
You know the right answer?
Based on the results of your experiment (see graph below), write a conclusion.
Steps
Restate...
Restate...
Questions in other subjects:



Mathematics, 27.01.2021 21:10


Mathematics, 27.01.2021 21:10



Mathematics, 27.01.2021 21:10

Mathematics, 27.01.2021 21:10

Mathematics, 27.01.2021 21:10



